Effect of age on chemical element contents in female thyroid investigated by some nuclear analytical methods
Abstract
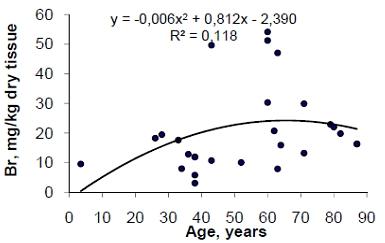
A prevalence of thyroid dysfunction is higher in the elderly as compared to the younger population. An excess or deficiency of chemical element contents in thyroid may play an important role in goitro- and carcinogenesis of gland. The variation with age of the mass fraction of twenty chemical elements (Ag, Br, Ca, Cl, Co, Cr, Cu, Fe, Hg, I, K, Mg, Mn, Na, Rb, Sb, Sc, Se, Sr, and Zn) in intact (normal) thyroid of 33 females (mean age 54.5 years, range 3.5-87) was investigated by energy dispersive X-ray fluorescent analysis and instrumental neutron activation analysis with high resolution spectrometry of short- and long-lived radionuclides. This work revealed that there is an increase in Br, Ca, Co, Fe, Rb, Sb, Se, and Zn mass fraction, as well as a decrease in Cl mass fraction in the normal thyroid of female during a lifespan. Therefore, a goitrogenic and carcinogenic effect of inadequate Br, Ca, Co, Fe, Rb, Sb, Se, and Zn level in the thyroid of old females with increasing age may be assumed.
References
2. Mitrou P, Raptis SA, Dimitriadis G. Thyroid disease in older people. Maturitas. 2011; 70: 5-9.
3. Kwong N, Medici M, Angell TE, Liu X, Marqusee E, Cibas ES, et al. The influence of patient age on thyroid nodule formation, multinodularity, and thyroid cancer risk. J Clin Endocrinol Metab. 2015; 100(12): 434-440.
4. Mazzaferri E. Management of a solitary thyroid nodule. NEJM. 1993; 328: 553-559.
5. Smailyte G, Miseikyte-Kaubriene E, Kurtinaitis J. Increasing thyroid cancer incidence in Lithuania in 1978-2003. BMC Cancer. 2006; 11(6): 284.
6. Olinski R, Siomek A, Rozalski R, Gackowski D, Foksinski M, Guz J, et al. Oxidative damage to DNA and antioxidant status in aging and age-related diseases. Acta Biochim Pol. 2007; 54: 11-26.
7. Minelli A, Bellezza I, Conte C, Culig Z. Oxidative stress-related aging: A role for prostate cancer? Biochim Biophys Acta. 2009; 1795: 83-91.
8. Klaunig JE, Kamendulis LM, Hocevar BA. Oxidative stress and oxidative damage in carcinogenesis. Toxicol Pathol. 2010; 38: 96-109.
9. Järup L. Hazards of heavy metal contamination. Br Med Bull. 2003; 68: 167-182.
10. Zaichick V, Zaichick S. Role of zinc in prostate cancerogenesis. In: Anke M, et al, eds. Mengen und Spurenelemente, 19 Arbeitstagung. Jena, Friedrich-Schiller-Universität, 1999: 104-115.
11. Zaichick V. INAA and EDXRF applications in the age dynamics assessment of Zn content and distribution in the normal human prostate. J Radioanal Nucl Chem. 2004; 262: 229-234.
12. Zaichick V. Medical elementology as a new scientific discipline. J Radioanal Nucl Chem. 2006; 269: 303-309.
13. Toyokuni S. Molecular mechanisms of oxidative stress-induced carcinogenesis: from epidemiology to oxygenomics. IUBMB Life. 2008; 60: 441-447.
14. Gupte A, Mumper RJ. Elevated copper and oxidative stress in cancer cells as a target for cancer treatment. Cancer Treat Rev. 2009; 35: 32-46.
15. Lee JD, Wu SM, Lu LY, Yang YT, Jeng SY. Cadmium concentration and metallothionein expression in prostate cancer and benign prostatic hyperplasia of humans. Taiwan Yizhi. 2009; 108: 554-559.
16. Zaichick V, Tsyb A, Vtyurin BM. Trace elements and thyroid cancer. Analyst. 1995; 120: 817-821.
17. Zaichick V, Choporov Yu. Determination of the natural level of human intra-thyroid iodine by instrumental neutron activation analysis. J Radioanal Nucl Chem. 1996; 207(1): 153-161.
18. Zaichick V, Zaichick S. Normal human intrathyroidal iodine. Sci Total Environ. 1997; 206(1): 39-56.
19. Zaichick V. Iodine excess and thyroid cancer. J Trace Elem Exp Med. 1998; 11(4): 508-509.
20. Zaichick V. In vivo and in vitro application of energy-dispersive XRF in clinical investigations: experience and the future. J Trace Elem Exp Med. 1998; 11: 509-510.
21. Zaichick V, Iljina T. Dietary iodine supplementation effect on the rat thyroid 131I blastomogenic action. In: Anke M, et al, eds. Die Bedentung der Mengen- und Spurenelemente. 18. Arbeitstangung. Jena, Friedrich-Schiller-Universität, 1998: 294-306.
22. Zaichick V, Zaichick S. Energy-dispersive X-ray fluorescence of iodine in thyroid puncture biopsy specimens. J Trace Microprobe Tech. 1999; 17: 219-232.
23. Zaichick V. Human intrathyroidal iodine in health and non-thyroidal disease. In: Abdulla M, et al, eds. New aspects of trace element research. London and Tokyo, Smith-Gordon and Nishimura, 1999: 114-119.
24. Zaichick V. Relevance of and potentiality for in vivo intrathyroidal iodine determination. In: Yasumura S, et al, eds. In vivo body composition studies. Ann NY Acad Sci. 2000; 904: 630-632.
25. Zhu H, Wang N, Zhang Y, Wu Q, Chen R, Gao J, et al. Element contents in organs and tissues of Chinese adult men. Health Phys. 2010; 98(1):61-73.
26. Vlasova ZA. Dynamics of trace element contents in thyroid gland in connection with age and atherosclerosis [in Russian]. Proceedings of the Leningrad Institute of Doctor Advanced Training 1969; 80: 135-144.
27. Salimi J, Moosavi K, Vatankhah S, Yaghoobi A. Investigation of heavy trace elements in neoplastic and non-neoplastic human thyroid tissue: A study by proton - induced X-ray emissions. Iran J Radiat Res. 2004; 1(4): 211-216.
28. Boulyga SF, Zhuk IV, Lomonosova EM, Kanash NV, Bazhanova NN. Determination of microelements in thyroids of the inhabitants of Belarus by neutron activation analysis using the k0-method. J Radioanal Nucl Chem. 1997; 222: 11-14.
29. Reddy SB, Charles MJ, Kumar MR, Reddy BS, Anjaneyulu Ch, Raju GJN, et al. Trace elemental analysis of adenoma and carcinoma thyroid by PIXE method. Nucl Instrum Methods Phys Res B. 2002; 196: 333-339.
30. Woodard HQ, White DR. The composition of body tissues. Brit J Radiol. 1986; 708: 1209-1218.
31. Katoh Y, Sato T, Yamamoto Y. Determination of multielement concentrations in normal human organs from the Japanese. Biol Trace Elem Res. 2002; 90(1-3): 57-70.
32. Tipton IH, Cook MJ. Trace elements in human tissue. Part II. Adult subjects from the United States. Health Phys. 1963; 9: 103-145.
33. Ataulchanov IA. Age-related changes of manganese, cobalt, coper, zinc, and iron contents in the endocrine glands of females [in Russian]. Problemy Endocrinologii. 1969; 15(2): 98-102.
34. Neimark II, Timoschnikov VM. Development of carcinoma of the thyroid gland in person residing in the focus of goiter endemic [in Russian]. Problemy Endocrinilogii. 1978; 24(3): 28-32.
35. Zabala J, Carrion N, Murillo M, Quintana M, Chirinos J, Seijas N, et al. Determination of normal human intrathyroidal iodine in Caracas population. J Trace Elem Med Bio. 2009; 23: 9-14.
36. Forssen A. Inorganic elements in the human body. Ann Med Exp Biol Fenn. 1972; 50: 99-162.
37. Kortev AI, Dontsov GI, Lyascheva AP. Bio-elements in human pathology [in Russian]. Sverdlovsk, Middle-Ural Publishing-House, 1972.
38. Soman SD, Joseph KT, Raut SJ, Mulay CD, Parameshwaran M, Panday VK, et al. Studies of major and trace element content in human tissues. Health Phys. 1970; 19: 641-656.
39. Teraoka H. Distribution of 24 elements in the internal organs of normal males and the metallic workers in Japan. Arch Environ Health. 1981; 36: 155-165.
40. Boulyga SF, Becker JS, Malenchenko AF, Dietze H-J. Application of ICP-MS for multielement analysis in small sample amounts of pathological thyroid tissue. MCA. 2000; 134: 215-222.
41. Fuzailov Yu M. Reaction of human and animal thyroids in the conditions of antimony sub-region of the Fergana valley [in Russian]. In: IX All-Union Conference on Trace Elements in Biology. Kishinev, 1981: 58-62.
42. Kvicala J, Havelka J, Zeman J, Nemec J. Determination of some trace elements in the thyroid gland by INAA. J Radioanal Nucl Chem. 1991; 149: 267-274.
43. Zaichick V. Sampling, sample storage and preparation of biomaterials for INAA in clinical medicine, occupational and environmental health. In: Harmonization of health-related environmental measurements using nuclear and isotopic techniques. Vienna, IAEA, 1997: 123-133.
44. Zaichick V. Losses of chemical elements in biological samples under the dry ashing process [in Russian]. Trace Elem Med. 2004; 5: 17-22.
45. Zaichick V, Zaichick S. INAA applied to halogen (Br and I) stability in long-term storage of lyophilized biological materials. J Radioanal Nucl Chem. 2000; 244: 279-281.
46. Zaichick V, Zaichick S. Instrumental effect on the contamination of biomedical samples in the course of sampling. J Anal Chem. 1996; 51(12): 1200-1205.
47. Zaichick V, Tsislyak Yu V. A simple device for bio-sample lyophilic drying [in Russian]. Laboratornoe Delo. 1978; 2: 109-110.
48. Zaichick V, Tsislyak Yu V. A modified adsorptive and cryogenic lyophilizer for biosample concentrations [in Russian]. Laboratornoe Delo. 1981; 2: 100-101.
49. Zaichick V, Zaichick S. A search for losses of chemical elements during freeze-drying of biological materials. J Radioanal Nucl Chem. 1997; 218: 249-253.
50. Zaichick S, Zaichick V. Method and portable facility for energy-dispersive X-ray fluorescent analysis of zinc content in needle-biopsy specimens of prostate. X-Ray Spectrom. 2010; 39: 83-89.
51. Zaichick S, Zaichick V. The Br, Fe, Rb, Sr, and Zn content and interrelation in intact and morphologic normal prostate tissue of adult men investigated by energy dispersive X-ray fluorescent analysis. X-Ray Spectrom. 2011; 40: 464-469.
52. Zaichick V. Applications of synthetic reference materials in the medical Radiological Research Centre. Fresen J Anal Chem. 1995; 352: 219-223.
53. Zaichick S, Zaichick V. The effect of age and gender on 37 chemical element contents in scalp hair of healthy humans. Biol Trace Elem Res. 2010; 134: 41-54.
54. Zaichick S, Zaichick V. The effect of age on Ag, Co, Cr, Fe, Hg, Sb, Sc, Se, and Zn contents in intact human prostate investigated by neutron activation analysis. J Appl Radiat Isot. 2011; 69: 827-833.
55. Korelo AM, Zaichick V. Software to optimize the multielement INAA of medical and environmental samples [in Russian]. In: Activation analysis in environment protection. Dubna, Joint Institute for Nuclear Research, 1993: 326-332.
56. Katoh Y, Sato T, Yamamoto Y. Determination of multielement concentrations in normal human organs from the Japanese. Biol Trace Elem Res. 2002; 90: 57-70.
57. Schroeder HA, Tipton IH, Nason AP. Trace metals in man: strontium and barium. J Chron Dis. 1972; 25: 491-517.
58. Pavelka S. Radiometric determination of thyrotoxic effects of some xenobiotics. Rad Applic. 2016; 1: 155-158.
59. Legrand G, Humez S, Slomianny C, Dewailly E, Vanden Abeele F, Mariot P, et al. Ca2+ pools and cell growth. Evidence for sarcoendoplasmic Ca2+-ATPases 2B involvement in human prostate cancer cell growth control. J Biol Chem. 2001; 276(50): 47608-47614.
60. Munarov L. Calcium signalling and control of cell proliferation by tyrosine kinase receptors. Int J Mol Med. 2002; 10: 671-676.
61. Prevarskaya N, Skryma R, Shuba Y. Ca2+ homeostasis in apoptotic resistance of prostate cancer cells. Biochem Biophys Res Commun. 2004; 322: 1326-1335.
62. Capiod T, Shuba Y, Skryma R, Prevarskaya N. Calcium signalling and cancer cell growth. Calcium signalling and disease. Subcell Biochem. 2007; 45: 405-427.
63. Roderick HL, Cook SJ. Ca2+ signalling checkpoints in cancer: remodelling Ca2+ for cancer cell proliferation and survival. Nat Rev Cancer. 2008; 8: 361-375.
64. Flourakis M, Prevarskaya N. Insights into Ca2+ homeostasis of advanced prostate cancer cells. Biochim Biophys Acta. 2009; 1793: 1105-1109.
65. Feng M, Grice DM, Faddy HM, Nguyen N, Leitch S, Wang Y, et al. Store-independent activation of Orai1 by SPCA2 in mammary tumors. Cell. 2010; 143: 84-98.
66. Yang H, Zhang Q, He J, Lu W. Regulation of calcium signaling in lung cancer. J Thorac Dis. 2010; 2(1): 52-56.
67. McAndrew D, Grice DM, Peters AA, Davis FM, Stewart T, Rice M, et al. ORAI1-mediated calcium influx in lactation and in breast cancer. Mol Cancer Ther. 2011; 10(3): 448-460.
68. Zaichick S, Zaichick V. INAA application in the age dynamics assessment of Br, Ca, Cl, K, Mg, Mn, and Na content in the normal human prostate. J Radioanal Nucl Chem. 2011; 288: 197-202.
69. Zaichick V, Nosenko S, Moskvina I. The effect of age on 12 chemical element contents in intact prostate of adult men investigated by inductively coupled plasma atomic emission spectrometry. Biol Trace Elem Res. 2012; 147: 49-58.
70. Feng MY, Rao R. New insights into store-independent Ca(2+) entry: secretory pathway calcium ATPase 2 in normal physiology and cancer. Int J Oral Sci. 2013; 5: 71-74.
71. Zaichick V, Zaichick S. INAA application in the assessment of chemical element mass fractions in adult and geriatric prostate glands. J Appl Radiat Isot. 2014; 90: 62-73.
72. Zaichick V, Zaichick S. Determination of trace elements in adults and geriatric prostate combining neutron activation with inductively coupled plasma atomic emission spectrometry. Open J Biochem. 2014; 1: 16-33.
73. Pavithra V, Sathisha TG, Kasturi K, Mallika DS, Amos SJ, Ragunatha S, et al. Serum levels of metal ions in female patients with breast cancer. J Clin Diagn Res. 2015; 9: BC25-BC27.
74. Zaichick V, Zaichick S, Davydov G. Differences between chemical element contents in hyperplastic and nonhyperplastic prostate glands investigated by neutron activation analysis. Biol Trace Elem Res. 2015; 164: 25-35.
75. Zaichick S, Zaichick V. Prostatic tissue level of some androgen dependent and independent trace elements in patients with benign prostatic hyperplasia. Androl Gynecol Curr Res. 2015; 3: 3.
76. Zaichick V, Zaichick S. The bromine, calcium, potassium, magnesium, manganese, and sodium contents in adenocarcinoma of human prostate gland. JHOR. 2016; 2: 1-12.
77. Zaichick V, Zaichick S. Variations in concentration and distribution of several androgen-dependent and -independent trace elements in nonhyperplastic prostate gland tissue throughout adulthood. J Androl Gynaecol. 2016; 4(1): 1-10.
78. Zaichick V, Zaichick S. Prostatic tissue levels of 43 trace elements in patients with prostate adenocarcinoma. CCO. 2016; 5(1): 79-94.
79. Zaichick V, Zaichick S. Levels of 43 trace elements in hyperplastic prostate tissues. BJMMR. 2016; 15(2): 1-12.
80. Zaichick V, Zaichick S. Prostatic tissue level of some major and trace elements in patients with BPH. J Nephrol Urol. 2016; 3(1): 025.
81. Zaichick V, Zaichick S. Age-related changes in concentration and histological distribution of Br, Ca, Cl, K, Mg, Mn, and Na in nonhyperplastic prostate of adults. EJBMSR. 2016; 4(2): 31-48.
82. Zaichick V, Zaichick S. Age-related changes in concentration and histological distribution of 18 chemical elements in nonhyperplastic prostate of adults. WJPMR. 2016; 2(4): 5-18.
83. Zaichick V, Zaichick S. The comparison between the contents and interrelationships of 17 chemical elements in normal and cancerous prostate gland. J Prostate Cancer. 2016; 1(1): 105.
84. Zaichick V, Zaichick S, Rossmann M. Intracellular calcium excess as one of the main factors in the etiology of prostate cancer. AIMS Mol Sci. 2016; 3(4): 635-647.
85. Sunderman FW. Mechanism of metal carcinogenesis. Biol Trace Elem Res. 1979; 1: 63-86.
86. Snow ET. Metal carcinogenesis: mechanistic implications. Pharmacol Ther. 1992; 53: 31-65.
87. Toyokuni S. Role of iron in carcinogenesis: cancer as a ferrotoxic disease. Cancer Sci. 2009; 100: 9-16.
88. Martinez-Zamudio R, Ha HC. Environmental epigenetics in metal exposure. Epigenetics. 2011; 6: 820-827.
89. Tokar EJ, Benbrahim-Tallaa L, Waalkes MP. Metal ions in human cancer development. Met Ions Life Sci. 2011; 8: 375-401.
90. Tchounwou PB, Yedjou CG, Patlolla AK, Sutton DJ. Heavy metal toxicity and the environment. EXS. 2012; 101: 133-164.
91. Koedrith P, Kim H, Weon JI, Seo YR. Toxicogenomic approaches for understanding molecular mechanisms of heavy metal mutagenicity and carcinogenicity. Int J Hyg Environ Health. 2013; 216: 587-598.
92. Tabrez S, Priyadarshini M, Priyamvada S, Khan MS, Na A, Zaidi SK, et al. Gene-environment interactions in heavy metal and pesticide carcinogenesis. Mutat Res. 2014; 760: 1-9.
93. Zaichick V, Zaichick S. Role of zinc in prostate cancerogenesis. In: Anke M, et al, eds. Mengen und Spurenelemente. 19. Arbeitstagung. Jena, Friedrich-Schiller-Universität, 1999: 104-115.
94. Zaichick V. INAA and EDXRF applications in the age dynamics assessment of Zn content and distribution in the normal human prostate. J Radioanal Nucl Chem. 2004; 262: 229-234.
95. Zaichick S, Zaichick V. The effect of age on Ag, Co, Cr, Fe, Hg, Sb, Sc, Se, and Zn contents in intact human prostate investigated by neutron activation analysis. J Appl Radiat Isot. 2011; 69: 827-833.
96. Zaichick S, Zaichick V, Nosenko S, Moskvina I. Mass fractions of 52 trace elements and zinc trace element content ratios in intact human prostates investigated by inductively coupled plasma mass spectrometry. Biol Trace Elem Res. 2012; 149: 171-183.
97. Zaichick V, Zaichick S. Use of INAA and ICP-MS for the assessment of trace element mass fractions in adult and geriatric prostate. J Radioanal Nucl Chem. 2014; 301: 383-397.
98. Zaichick V, Zaichick S. Age-related histological and zinc content changes in adult nonhyperplastic prostate glands. Age. 2014; 36: 167-181.
99. Zaichick V, Zaichick S. Differences and relationships between morphometric parameters and zinc content in nonhyperplastic and hyperplastic prostate glands. BJMMR. 2015; 8(8): 692-706.
100. Zaichick V, Zaichick S. Trace element contents in adenocarcinoma of human prostate investigated by energy dispersive X-ray fluorescent analysis. J Adenocarcinoma. 2016; 1(1): 1-7.
101. Zaichick V, Zaichick S. Trace element contents in adenocarcinoma of the human prostate gland investigated by neutron activation analysis. CROOA. 2016; 1(1): 1-10.
102. Zaichick V, Zaichick S. Variations in concentration and histological distribution of Ag, Co, Cr, Fe, Hg, Rb, Sb, Sc, Se, and Zn in nonhyperplastic prostate gland throughout adulthood. J Cell Mol Biol. 2016; 2(1): 011.
103. Zaichick V, Zaichick S. Age-related changes in concentration and histological distribution of 54 trace elements in nonhyperplastic prostate of adults. Int Arch Urol Complic. 2016; 2(2): 019.
104. Zaichick V, Zaichick S, Wynchank S. Intracellular zinc excess as one of the main factors in the etiology of prostate cancer. J Analytic Oncol. 2016; 5(3): 124-131.
105. Salnikow K, Zhitkovich A. Genetic and epigenetic mechanisms in metal carcinogenesis and cocarcinogenesis: nickel, arsenic, and chromium. Chem Res Toxicol. 2008; 21: 28-44.
106. Chervona Y, Arita A, Costa M. Carcinogenic metals and the epigenome: understanding the effect of nickel, arsenic, and chromium. Metallomics. 2012; 4: 619-627.
107. Aaseth J, Frey H, Glattre E, Norheim G, Ringstad J, Thomassen Y. Selenium concentrations in the human thyroid gland. Biol Trace Elem Res. 1990; 24(2-3): 147-152.

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.



