Pseudomonas species from cattle dung producing extended spectrum and metallo beta-lactamases
Abstract
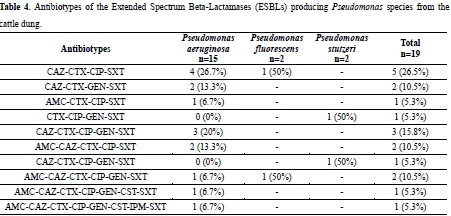
Indiscriminate use of antibiotics in livestock contributes to emergence of antimicrobial resistance in pathogens co-habiting the gastro-intestinal tract of animals. This study was to determine the Extended Spectrum Beta-Lactamase (ESBL) and Metallo-Beta-Lactamase (MBL) production in Pseudomonas species from cattle fecal samples. Cattle dungs were collected from the University of Ibadan Cattle Ranch and the Pseudomonas species isolated using Pseudomonas Base Agar with Pseudomonas CN Selective Supplement were identified using standard tests. Phenotypic detection of ESBL and MBL was by double disk synergy test and Ethylene Di-amine Tetra Acetic Acid Combined Disk Test respectively. Antibiotics susceptibility tests was done using the disc diffusion technique against ten antibiotics. A total of 144 Pseudomonas species were isolated and identified as P. aeruginosa (71.5%), P. fluorescens (19.4%) and P. stutzeri (9.1%) and 19 (37.1%) produced ESBL including P. aeruginosa (15), P. fluorescens (2) and P. stutzeri (2) while, one (6.7%) ESBL P. aeruginosa produced MBL. All the ESBL producers were resistant to cefotaxime and trimethoprim; resistance of P. aeruginosa to ciprofloxacin was 93.3% and to ceftazidime was 80.0%, while it was 13.3% (colistin) and 6.7% (imipenem). The ESBL producing P. fluorescens were resistant to ceftazidime, ciprofloxacin and trimethoprim, likewise, the ESBL producing P. stutzeri showed resistance to gentamicin, ciprofloxacin and trimethoprim. The production of ESBL and MBL observed among the Pseudomonas species in this study with high level of resistance to some antibiotics portend public health risk, hence a need for caution in the use of antibiotics in animal husbandry.
Downloads
References
2. Woolhouse M, Ward M, Van Bunnik B, Farrar J. Antimicrobial resistance in humans, livestock and the wider environment. Phil Trans Royal Soc B Lond Ser B, Biol Sci. 2015; 370: 1-7.
3. Nordmann P, Naas T, Poirel L. Global spread of carbapenemase-producing Enterobacteriaceae. Emerg Infec Dis. 2011; 17: 1791-1798.
4. Ahmad M, Hassan M, Khalid A, Tariq I, Asad M, Samad A, et al. Prevalence of extended spectrum β-lactamase and antimicrobial susceptibility pattern of clinical isolates of Pseudomonas from patients of Khyber Pakhtunkhwa, Pakistan. BioMed Res Int. 2016, 2016: 2-8.
5. Capita R, Alonso-Calleja C. Antibiotic-resistant bacteria: A challenge for the food industry antibiotic-resistant bacteria. Crit Rev Food Sci Nutr. 2016; 53: 11-48.
6. Landers TF, Cohen B, Wittum TE, Larson EL. A review of antibiotic use in food animals: Perspective, policy, and potential. Publ Hlth Reports. 2012; 127: 4-22.
7. Marshall BM, Levy SB. Food Animals and antimicrobials: Impacts on human health. Clin Microbiol Rev. 2011; 24: 718-733.
8. Paterson DL, Bonomo RA. Extended-spectrum β-lactamases: A clinical update. Clin Microbiol Rev. 2005; 18: 657-686.
9. Vijay M, Urekar AD, Goel Insan N. Extended Spectrum Beta-Lactamases, Metallo Beta-Lactamases and AMP C detection in multidrug resistant Pseudomonas aeruginosa and pan drug resistant Pseudomonas aeruginosa isolated in Tertiary Care Hospital. Int J Curr Microbiol Appl Sci. 2014; 3(11): 489-492.
10. Walsh TR, Toleman MA, Poirel L, Nordmann P. Metallo-beta-lactamases: the quiet before the storm? Clin Microbiol Rev. 2005; 18: 306-325.
11. Pitout JD, Gregson L, Poirel JA, McClure P, Church DL. Detection of Pseudomonas aeruginosa producing metallo-beta-lactamases in a large centralized laboratory. J Clin Microbiol. 2005; 43: 3129-3135.
12. Kathiravan T, Marykala J, Sundaramanickam A, Kumaresan S, Balasubramanian T. Studies on nutritional requirements of Pseudomonas aeruginosa for lipase production. Adv Appl Sci Res. 2012; 3(1): 591-598.
13. Cheesbrough M. District laboratory practice for tropical countries (Part 2). Cambridge University Press. 2006: 180-197.
14. Clinical Laboratory Standard Institute. Performance standards for antimicrobial susceptibility testing. 28th edn. CLSI supplement M100. Wayne, PA: Clinical and Laboratory Standards Institute; 2018.
15. Drieux L, Brossier F, Sougakoff W, Jarlier V. Phenotypic detection of extended spectrum beta-lactamase production in Enterobacteriaceae: review and bench guide. Clin Microbiol Infect. 2008; 1: 190-103.
16. Galani I, Rekatsina PD, Hatzaki D, Plachouras D, Souli M, Giamarellou H. Evaluation of different laboratory tests for the detection of metallo-beta-lactamase production in Enterobacteriaceae. J Antimicrob Chemoth. 2008; 61(3): 548-553.
17. Adamu MT, Jaafaru MI. Prevalence and antimicrobial sensitivity profile of Salmonella typhimurium and Escherichia coli O157:H7 In cattle feces, drainage and waste water at Gombe abattoir environment in Nigeria. Int J Adv Sci Eng Tech. 2015; 3(4): 30-34.
18. Asikong BE, Udensi OU, Epoke J, Eja EM, Antai EE. Microbial analysis and Biogas yield of water hyacinth, cow dung and poultry dopping fed anaerobic digesters. Brit J Appl Sci Techn. 2014; 4(4): 650-661.
19. Godambe T, Fulekar MH. Cow dung bacteria offer an effective bioremediation for hydrocarbon benzene. Int J Biotech Trends Techn. 2016; 6(3): 13-20.
20. Umanu G, Owoseni RA. Effects of abattoir effluent on microbial degradation of diesel oil in tropical agricultural soil. Pac J Sci Tech. 2013; 14(1): 604-612.
21. Ejikeugwu C, Esimone C, Iroha I, Ugwu C, Ezeador C. Phenotypic detection of AmpC beta-lactamase among anal Pseudomonas aeruginosa isolates in a Nigerian abattoir. Arch Clin Microbiol. 2016; 7(2): 1-5.
22. Odumosu BT, Ajetunmobi O, Dada-Adegbola H, Odutayo I. Antibiotic susceptibility pattern and analysis of plasmid profiles of Pseudomonas aeruginosa from human, animal and plant sources. Springer Plus. 2016; 5(1): 1381.
23. Wamala SP, Mugimba KK, Mutoloki S, Mdegela R, Byarugaba DK, Sorum H. Occurrence and antibiotic susceptibility of fish bacteria isolated from Oreochromis niloticus (Nile tilapia) and Clarias gariepinus (African catfish) in Uganda. Fisher Aqua Sci. 2018; 21: 6.
24. Kummerer K. Resistance in the environment. J Antimicrob Chemoth. 2004; 54: 311-320.
25. Igbinosa IH, Nwodo UU, Sosa A, Tom M, Okoh AI. Commensal Pseudomonas species isolated from wastewater and freshwater milieus in the Eastern Cape Province, South Africa, as reservoir of antibiotic resistant determinants. Int J Environ Res Pub Hlth. 2012; 9: 2537-2549.
26. Falodun OI, Morankinyo YM, Fagade OE. Determination of water quality and detection of extended spectrum beta-lactamase producing Gram-negative bacteria in selected rivers located in Ibadan, Nigeria. Jord J Biol Sci. 2018; 11: 107-112.
27. Begum S, Abdus-salam MD, Faisal Alarm KH, Begum N, Hassan P, AshrafulHaq J. Detection of extended spectrum β-lactamase in Pseudomonas species isolated from two tertiary care hospitals in Bangladesh. BMC Res Not. 2013; 6: 7.
28. Ruth AA, Damian CO, Romanus II, Charles OE. Antimicrobial resistance status and prevalence rates of extended spectrum beta-lactamase producers isolated from a mixed human population. Bos J Bas Med Sci. 2011; 11(2): 91-96.
29. Olga P, Apostolos V, Alexis G, George V, Athena M. Antibiotic resistance profiles of Pseudomonas aeruginosa isolated from various Greek aquatic environments. FEMS Microbiol Ecol. 2016; 92(5): 1-9.
30. Umadevi S, Joseph NM, Kumari K, Easow JM, Kumar S, Stephen, et al. Detection of extended spectrum beta lactamases, ampc beta-lactamases and metallobetalactamases in clinical isolates of ceftazidime resistant Pseudomonas aeruginosa. Braz J Microbiol. 2011; 42(4): 1284-1288.
31. Hocquet D, Plésiat P, Dehecq B, Mariotte P, Talon D, Xavier B. Nationwide investigation of extended-spectrum β-lactamases, metallo-β-lactamases, and extended-spectrum oxacillinases produced by ceftazidime-resistant Pseudomonas aeruginosa strains in France. Antimicrob Agents Chemoth. 2010; 54(8): 3512-3515.
32. Akinduti PA, Ejilude O, Motayo BO, Adeyokinu AF. Emerging multidrug resistant AmpC betalactamase and carbapenemase enteric isolates in Abeokuta, Nigeria. Nat Sci. 2012; 10: 7074.
33. Franklin C, Liolios L, Peleg A. Phenotypic detection of carbapenem susceptible metallo-β-lactamase producing Gram negative bacilli in the clinical laboratory. J Clin Microbiol. 2006; 44: 3139-3144.
34. Oberoi L, Singh N, Sharma P, Aggarwal A. Extended spectrum beta-lactamase, metallo beta-lactamas and ampc β-lactamases producing superbugs - Havoc in the intensive care units of Punjab India. J Clin Diag Res. 2013; 7: 70-83.
35. Ayatollahi J, Yousefi Y, Shahcheraghi SH. Study of drug resistance of Pseudomonas aeruginosa in Yazd, Iran, during 2015-2016. Int J Infect. 2018; 5(3): e68749.
36. Elhariri M, Hamza D, Elhelw R, Doegham SM. Extended-spectrum beta-lactamase-producing Pseudomonas aeruginosa in camel in Egypt: potential human hazard. Ann Clin Microbiol Antimicrob. 2017; 16: 21-31.
37. Rabiu AG, Falodun OI. Multi-drug resistant Pseudomonas species isolated from the wastewater of an abattoir in Ibadan, Nigeria. J App Life Sci Int. 2017; 13(1): 1-9.
38. Geser N, Stephan R, Kuhnert P, Zbinden R, Kaeppeli U, Cernela N, et al. Fecal carriage of extended-spectrum betalactamase-producing Enterobacteriaceae in swine and cattle at slaughter in Switzerland. J Food Protect. 2011; 74: 446-449.
39. Livermore DM. Multiple mechanisms of antimicrobial resistance in Pseudomonas aeruginosa: Our worst nightmare? CID Antimicrob Resist. 2004; 34: 534-664.
40. Shahcheraghi F, Nikbin V, Feizabadi MM. Prevalence of ESBLs genes among multidrug-resistant isolates of Pseudomonas aeruginosa isolated from patients in Tehran. Microbiol Drug Res. 2009; 15: 37-39.
41. Chen Z, Niu H, Chen G, Li M, Li M, Zhou Y. Prevalence of extended spectrum beta-lactamases producing Pseudomonas aeruginosa isolates from different wards in a Chinese Teaching Hospital. Int J Clin Exp Med. 2015; 8: 19400-19405.

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.




